Committed to Evidence-Based Medicine, Gold-Standard Science and Common Sense Approach to Patient, Subject and Public Safety
Over the last 20+ years, BlueCloudX® has developed “National and International Industry Standards Through Crowd Sourcing”. These global standards have now proven to minimize fraud, waste, abuse, and redundancies for healthcare and clinical research professionals, national and international industry stakeholders, including but not limited to governments, research sites, hospitals, pharma, medical device, biotech, digital sponsors, universities as well as technology companies. The standards minimize data variance in clinical trials, with an overarching goal to develop a globally interconnected system that provides access to patients no matter race, religion, socio-economic status, political affiliation or geographical location. A global system that leaves no patient behind®.
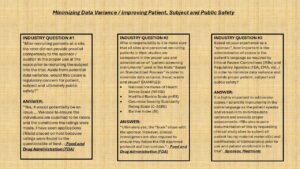
“Synopsis from Regulatory Agencies, Sponsors, CROs, Universities, IRBs and other Life and Health Sciences Industry Stakeholders – How Following Proper Standards Can Minimize and Ultimately Eliminate Warning Letters from the FDA”
Synopsis-FollowingProperStandards
=========================================================================================================================
Below are some of the industry standards which have now become globally accepted by over 2.6+ million healthcare and clinical research professionals from over 550,000 organizations in 193 countries:
DOWNLOADABLE “AUDIT READY” GLOBALLY STANDARDIZED TRAINING & CERTIFICATION PROCESSES FOR HEALTHCARE & CLINICAL RESEARCH PROFESSIONALS, SPONSORS, CROs, SITES, HOSPITALS AND ALL OTHER LIFE SCIENCES BASED STAKEHOLDERS
Self Manage Self Test (SMST)
- M-Appendix-M-0108-STANDARDS-ENGLISH- SelfManageSelfTest-SMST_v7.5
- M-Appendix-M-0108-STANDARDS-ARABIC- SelfManageSelfTest-SMST_v7.5
- M-Appendix-M-0108-STANDARDS-BULGARIAN- SelfManageSelfTest-SMST_v7.5
- M-Appendix-M-0108-STANDARDS-CHINESE TRADITIONAL- SelfManageSelfTest-SMST_v7.5
- M-Appendix-M-0108-STANDARDS-CROATIAN- SelfManageSelfTest-SMST_v7.5
- M-Appendix-M-0108-STANDARDS-CZECH- SelfManageSelfTest-SMST_v7.5
- M-Appendix-M-0108-STANDARDS-DANISH- SelfManageSelfTest-SMST_v7.5
- M-Appendix-M-0108-STANDARDS-DUTCH- SelfManageSelfTest-SMST_v7.5
- M-Appendix-M-0108-STANDARDS-FINNISH- SelfManageSelfTest-SMST_v7.5
- M-Appendix-M-0108-STANDARDS-FRENCH- SelfManageSelfTest-SMST_v7.5
- M-Appendix-M-0108-STANDARDS-GERMAN- SelfManageSelfTest-SMST_v7.5
- M-Appendix-M-0108-STANDARDS-GREEK- SelfManageSelfTest-SMST_v7.5
- M-Appendix-M-0108-STANDARDS-HEBREW- SelfManageSelfTest-SMST_v7.5
- M-Appendix-M-0108-STANDARDS-HUNGARIAN- SelfManageSelfTest-SMST_v7.5
- M-Appendix-M-0108-STANDARDS-ITALIAN- SelfManageSelfTest-SMST_v7.5
- M-Appendix-M-0108-STANDARDS-JAPANESE- SelfManageSelfTest-SMST_v7.5
- M-Appendix-M-0108-STANDARDS-KOREAN- SelfManageSelfTest-SMST_v7.5
- M-Appendix-M-0108-STANDARDS-LATVIAN- SelfManageSelfTest-SMST_v7.5
- M-Appendix-M-0108-STANDARDS-LITHUANIAN- SelfManageSelfTest-SMST_v7.5
- M-Appendix-M-0108-STANDARDS-MALAY- SelfManageSelfTest-SMST_v7.5
- M-Appendix-M-0108-STANDARDS-POLISH- SelfManageSelfTest-SMST_v7.5
- M-Appendix-M-0108-STANDARDS-PORTUGUESE- SelfManageSelfTest-SMST_v7.5
- M-Appendix-M-0108-STANDARDS-RUSSIAN- SelfManageSelfTest-SMST_v7.5
- M-Appendix-M-0108-STANDARDS-SERBIAN- SelfManageSelfTest-SMST_v7.5
- M-Appendix-M-0108-STANDARDS-SPANISH- SelfManageSelfTest-SMST_v7.5
- M-Appendix-M-0108-STANDARDS-SWEDISH- SelfManageSelfTest-SMST_v7.5
- M-Appendix-M-0108-STANDARDS-TAMIL- SelfManageSelfTest-SMST_v7.5
- M-Appendix-M-0108-STANDARDS-TURKISH- SelfManageSelfTest-SMST_v7.5
- M-Appendix-M-0108-STANDARDS-UKRAINIAN- SelfManageSelfTest-SMST_v7.5
- M-Appendix-M-0108-STANDARDS-VIETNAMESE- SelfManageSelfTest-SMST_v7.5
National Institutes of Health Stroke Scale (NIHSS)
- M-Appendix-M-0108-STANDARDS-ENGLISH- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-ARABIC- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-BULGARIAN- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-CHINESE SIMPLIFIED- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-CROATIAN- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-CZECH- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-DANISH- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-DUTCH- NationalInstitutesofHealthStrokeScale-NIHSS_v7.
- M-Appendix-M-0108-STANDARDS-FINNISH- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-FRENCH- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-GERMAN- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-GREEK- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-HEBREW- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-HINDI- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-HUNGARIAN- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-INDONESIAN- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-ITALIAN- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-JAPANESE- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-KAZAKH- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-KOREAN- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-LATVIAN- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-LITHUANIAN- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-MALAY- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-NORWEGIAN- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-POLISH- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-PORTUGUESE- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-ROMANIAN- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-RUSSIAN- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-SERBIAN- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-SPANISH- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-SWEDISH- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-TAMIL- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-THAI- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-TURKISH- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-UKRAINIAN- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
- M-Appendix-M-0108-STANDARDS-VIETNAMESE- NationalInstitutesofHealthStrokeScale-NIHSS_v7.5
Modified Rankin Scale (mRS)
- M-Appendix-M-0108-STANDARDS-ENGLISH- ModifiedRankinScale-mRS_v7.5
- M-Appendix-M-0108-STANDARDS-AFRIKAANS- ModifiedRankinScale-mRS_v7.5
- M-Appendix-M-0108-STANDARDS-ARABIC- ModifiedRankinScale-mRS_v7.5
- M-Appendix-M-0108-STANDARDS-BULGARIAN- ModifiedRankinScale-mRS_v7.5
- M-Appendix-M-0108-STANDARDS-CHINESE TRADITIONAL- ModifiedRankinScale-mRS_v7.5
- M-Appendix-M-0108-STANDARDS-CROATIAN- ModifiedRankinScale-mRS_v7.5
- M-Appendix-M-0108-STANDARDS-CZECH- ModifiedRankinScale-mRS_v7.5
- M-Appendix-M-0108-STANDARDS-DANISH- ModifiedRankinScale-mRS_v7.5
- M-Appendix-M-0108-STANDARDS-DUTCH- ModifiedRankinScale-mRS_v7.5
- M-Appendix-M-0108-STANDARDS-FINNISH- ModifiedRankinScale-mRS_v7.5
- M-Appendix-M-0108-STANDARDS-FRENCH- ModifiedRankinScale-mRS_v7.5
- M-Appendix-M-0108-STANDARDS-GERMAN- ModifiedRankinScale-mRS_v7.5
- M-Appendix-M-0108-STANDARDS-GREEK- ModifiedRankinScale-mRS_v7.5
- M-Appendix-M-0108-STANDARDS-HEBREW- ModifiedRankinScale-mRS_v7.5
- M-Appendix-M-0108-STANDARDS-HUNGARIAN- ModifiedRankinScale-mRS_v7.5
- M-Appendix-M-0108-STANDARDS-ITALIAN- ModifiedRankinScale-mRS_v7.5
- M-Appendix-M-0108-STANDARDS-JAPANESE- ModifiedRankinScale-mRS_v7.5
- M-Appendix-M-0108-STANDARDS-KAZAKH- ModifiedRankinScale-mRS_v7.5
- M-Appendix-M-0108-STANDARDS-KOREAN- ModifiedRankinScale-mRS_v7.5
- M-Appendix-M-0108-STANDARDS-LATVIAN- ModifiedRankinScale-mRS_v7.5
- M-Appendix-M-0108-STANDARDS-LITHUANIAN- ModifiedRankinScale-mRS_v7.5
- M-Appendix-M-0108-STANDARDS-MALAY- ModifiedRankinScale-mRS_v7.5
- M-Appendix-M-0108-STANDARDS-POLISH- ModifiedRankinScale-mRS_v7.5
- M-Appendix-M-0108-STANDARDS-PORTUGUESE- ModifiedRankinScale-mRS_v7.5
- M-Appendix-M-0108-STANDARDS-PUNJABI- ModifiedRankinScale-mRS_v7.5
- M-Appendix-M-0108-STANDARDS-ROMANIAN- ModifiedRankinScale-mRS_v7.5
- M-Appendix-M-0108-STANDARDS-RUSSIAN- ModifiedRankinScale-mRS_v7.5
- M-Appendix-M-0108-STANDARDS-SERBIAN- ModifiedRankinScale-mRS_v7.5
- M-Appendix-M-0108-STANDARDS-SPANISH- ModifiedRankinScale-mRS_v7.5
- M-Appendix-M-0108-STANDARDS-TAMIL- ModifiedRankinScale-mRS_v7.5
- M-Appendix-M-0108-STANDARDS-TURKISH- ModifiedRankinScale-mRS_v7.5
- M-Appendix-M-0108-STANDARDS-UKRAINIAN- ModifiedRankinScale-mRS_v7.5
- M-Appendix-M-0108-STANDARDS-URDU- ModifiedRankinScale-mRS_v7.5
- M-Appendix-M-0108-STANDARDS-VIETNAMESE- ModifiedRankinScale-mRS_v7.5
Columbia-Suicide Severity Rating Scale (C-SSRS)
- M-Appendix-M-0108-STANDARDS-ENGLISH- C-SSRS_v7.5
- M-Appendix-M-0108-STANDARDS-ARABIC- C-SSRS_v7.5
- M-Appendix-M-0108-STANDARDS-BULGARIAN- C-SSRS_v7.5
- M-Appendix-M-0108-STANDARDS-CHINESE TRADITIONAL- C-SSRS_v7.5
- M-Appendix-M-0108-STANDARDS-CROATIAN- C-SSRS_v7.5
- M-Appendix-M-0108-STANDARDS-CZECH- C-SSRS_v7.5
- M-Appendix-M-0108-STANDARDS-DANISH- C-SSRS_v7.5
- M-Appendix-M-0108-STANDARDS-DUTCH- C-SSRS_v7.5
- M-Appendix-M-0108-STANDARDS-ESTONIAN- C-SSRS_v7.5
- M-Appendix-M-0108-STANDARDS-FINNISH- C-SSRS_v7.5
- M-Appendix-M-0108-STANDARDS-FRENCH- C-SSRS_v7.5
- M-Appendix-M-0108-STANDARDS-GERMAN- C-SSRS_v7.5
- M-Appendix-M-0108-STANDARDS-GREEK- C-SSRS_v7.5
- M-Appendix-M-0108-STANDARDS-HEBREW- C-SSRS_v7.5
- M-Appendix-M-0108-STANDARDS-HUNGARIAN- C-SSRS_v7.5
- M-Appendix-M-0108-STANDARDS-ITALIAN- C-SSRS_v7.5
- M-Appendix-M-0108-STANDARDS-JAPANESE- C-SSRS_v7.5
- M-Appendix-M-0108-STANDARDS-KOREAN- C-SSRS_v7.5
- M-Appendix-M-0108-STANDARDS-LATVIAN- C-SSRS_v7.5
- M-Appendix-M-0108-STANDARDS-LITHUANIAN- C-SSRS_v7.5
- M-Appendix-M-0108-STANDARDS-MALAY- C-SSRS_v7.5
- M-Appendix-M-0108-STANDARDS-POLISH- C-SSRS_v7.5
- M-Appendix-M-0108-STANDARDS-PORTUGUESE- C-SSRS_v7.5
- M-Appendix-M-0108-STANDARDS-ROMANIAN- C-SSRS_v7.5
- M-Appendix-M-0108-STANDARDS-RUSSIAN- C-SSRS_v7.5
- M-Appendix-M-0108-STANDARDS-SERBIAN- C-SSRS_v7.5
- M-Appendix-M-0108-STANDARDS-SLOVAK- C-SSRS_v7.5
- M-Appendix-M-0108-STANDARDS-SPANISH- C-SSRS_v7.5
- M-Appendix-M-0108-STANDARDS-SWEDISH- C-SSRS_v7.5
- M-Appendix-M-0108-STANDARDS-TAMIL- C-SSRS_v7.5
- M-Appendix-M-0108-STANDARDS-TURKISH- C-SSRS_v7.5
- M-Appendix-M-0108-STANDARDS-UKRAINIAN- C-SSRS_v7.5
- M-Appendix-M-0108-STANDARDS-VIETNAMESE- C-SSRS_v7.5
Barthel Index (BI)
- M-Appendix-M-0108-STANDARDS-ENGLISH- Barthel Index_v7.5
- M-Appendix-M-0108-STANDARDS-ARABIC- Barthel Index_v7.5
- M-Appendix-M-0108-STANDARDS-BULGARIAN- Barthel Index_v7.5
- M-Appendix-M-0108-STANDARDS-CHINESE TRADITIONAL- Barthel Index_v7.5
- M-Appendix-M-0108-STANDARDS-CROATIAN- Barthel Index_v7.5
- M-Appendix-M-0108-STANDARDS-CZECH- Barthel Index_v7.5
- M-Appendix-M-0108-STANDARDS-DANISH- Barthel Index_v7.5
- M-Appendix-M-0108-STANDARDS-DUTCH- Barthel Index_v7.5
- M-Appendix-M-0108-STANDARDS-FINNISH- Barthel Index_v7.5
- M-Appendix-M-0108-STANDARDS-FRENCH- Barthel Index_v7.5
- M-Appendix-M-0108-STANDARDS-GERMAN- Barthel Index_v7.5
- M-Appendix-M-0108-STANDARDS-GREEK- Barthel Index_v7.5
- M-Appendix-M-0108-STANDARDS-HEBREW- Barthel Index_v7.5
- M-Appendix-M-0108-STANDARDS-HUNGARIAN- Barthel Index_v7.5
- M-Appendix-M-0108-STANDARDS-ITALIAN- Barthel Index_v7.5
- M-Appendix-M-0108-STANDARDS-JAPANESE- Barthel Index_v7.5
- M-Appendix-M-0108-STANDARDS-KOREAN- Barthel Index_v7.5
- M-Appendix-M-0108-STANDARDS-LATVIAN- Barthel Index_v7.5
- M-Appendix-M-0108-STANDARDS-LITHUANIAN- Barthel Index_v7.5
- M-Appendix-M-0108-STANDARDS-MALAY- Barthel Index_v7.5
- M-Appendix-M-0108-STANDARDS-POLISH- Barthel Index_v7.5
- M-Appendix-M-0108-STANDARDS-PORTUGUESE- Barthel Index_v7.5
- M-Appendix-M-0108-STANDARDS-PUNJABI- Barthel Index_v7.5
- M-Appendix-M-0108-STANDARDS-RUSSIAN- Barthel Index_v7.5
- M-Appendix-M-0108-STANDARDS-SERBIAN- Barthel Index_v7.5
- M-Appendix-M-0108-STANDARDS-SPANISH- Barthel Index_v7.5
- M-Appendix-M-0108-STANDARDS-SWEDISH- Barthel Index_v7.5
- M-Appendix-M-0108-STANDARDS-TAMIL- Barthel Index_v7.5
- M-Appendix-M-0108-STANDARDS-TURKISH- Barthel Index_v7.5
- M-Appendix-M-0108-STANDARDS-UKRAINIAN- Barthel Index_v7.5
- M-Appendix-M-0108-STANDARDS-VIETNAMESE- Barthel Index_v7.5
Fugl Meyer Arm/Leg Scales (FMA / FML)
- M-Appendix-M-0108-STANDARDS-ARABIC- Fugl-Meyer-Scale_v7.6
- M-Appendix-M-0108-STANDARDS-BULGARIAN- Fugl-Meyer-Scale_v7.6
- M-Appendix-M-0108-STANDARDS-CHINESE TRADITIONAL- Fugl-Meyer-Scale_v7.6
- M-Appendix-M-0108-STANDARDS-CROATIAN- Fugl-Meyer-Scale_v7.6
- M-Appendix-M-0108-STANDARDS-CZECH- Fugl-Meyer-Scale_v7.6
- M-Appendix-M-0108-STANDARDS-DANISH- Fugl-Meyer-Scale_v7.6
- M-Appendix-M-0108-STANDARDS-DUTCH- Fugl-Meyer-Scale_v7.6
- M-Appendix-M-0108-STANDARDS-ENGLISH- Fugl-Meyer-Scale_v7.6
- M-Appendix-M-0108-STANDARDS-FINNISH- Fugl-Meyer-Scale_v7.6
- M-Appendix-M-0108-STANDARDS-FRENCH- Fugl-Meyer-Scale_v7.6
- M-Appendix-M-0108-STANDARDS-GERMAN- Fugl-Meyer-Scale_v7.6
- M-Appendix-M-0108-STANDARDS-GREEK- Fugl-Meyer-Scale_v7.6
- M-Appendix-M-0108-STANDARDS-HEBREW- Fugl-Meyer-Scale_v7.6
- M-Appendix-M-0108-STANDARDS-HUNGARIAN- Fugl-Meyer-Scale_v7.6
- M-Appendix-M-0108-STANDARDS-ITALIAN- Fugl-Meyer-Scale_v7.6
- M-Appendix-M-0108-STANDARDS-JAPANESE- Fugl-Meyer-Scale_v7.6
- M-Appendix-M-0108-STANDARDS-KOREAN- Fugl-Meyer-Scale_v7.6
- M-Appendix-M-0108-STANDARDS-LATVIAN- Fugl-Meyer-Scale_v7.6
- M-Appendix-M-0108-STANDARDS-LITHUANIAN- Fugl-Meyer-Scale_v7.6
- M-Appendix-M-0108-STANDARDS-MALAY- Fugl-Meyer-Scale_v7.6
- M-Appendix-M-0108-STANDARDS-POLISH- Fugl-Meyer-Scale_v7.6
- M-Appendix-M-0108-STANDARDS-PORTUGUESE- Fugl-Meyer-Scale_v7.6
- M-Appendix-M-0108-STANDARDS-RUSSIAN- Fugl-Meyer-Scale_v7.6
- M-Appendix-M-0108-STANDARDS-SERBIAN- Fugl-Meyer-Scale_v7.6
- M-Appendix-M-0108-STANDARDS-SPANISH- Fugl-Meyer-Scale_v7.6
- M-Appendix-M-0108-STANDARDS-SWEDISH- Fugl-Meyer-Scale_v7.6
- M-Appendix-M-0108-STANDARDS-TAMIL- Fugl-Meyer-Scale_v7.6
- M-Appendix-M-0108-STANDARDS-TURKISH- Fugl-Meyer-Scale_v7.6
- M-Appendix-M-0108-STANDARDS-UKRAINIAN- Fugl-Meyer-Scale_v7.6
- M-Appendix-M-0108-STANDARDS-VIETNAMESE- Fugl-Meyer-Scale_v7.6
Rankin Focused Assessment (RFA)
- M-Appendix-M-0108-STANDARDS-ENGLISH- RFA-Rankin_v7.5
- M-Appendix-M-0108-STANDARDS-ARABIC- RFA-Rankin_v7.5
- M-Appendix-M-0108-STANDARDS-BULGARIAN- RFA-Rankin_v7.5
- M-Appendix-M-0108-STANDARDS-CHINESE TRADITIONAL- RFA-Rankin_v7.5
- M-Appendix-M-0108-STANDARDS-CROATIAN- RFA-Rankin_v7.5
- M-Appendix-M-0108-STANDARDS-CZECH- RFA-Rankin_v7.5
- M-Appendix-M-0108-STANDARDS-DANISH- RFA-Rankin_v7.5
- M-Appendix-M-0108-STANDARDS-DUTCH- RFA-Rankin_v7.5
- M-Appendix-M-0108-STANDARDS-FINNISH- RFA-Rankin_v7.5
- M-Appendix-M-0108-STANDARDS-FRENCH- RFA-Rankin_v7.5
- M-Appendix-M-0108-STANDARDS-GERMAN- RFA-Rankin_v7.5
- M-Appendix-M-0108-STANDARDS-GREEK- RFA-Rankin_v7.5
- M-Appendix-M-0108-STANDARDS-HEBREW- RFA-Rankin_v7.5
- M-Appendix-M-0108-STANDARDS-HUNGARIAN- RFA-Rankin_v7.5
- M-Appendix-M-0108-STANDARDS-ITALIAN- RFA-Rankin_v7.5
- M-Appendix-M-0108-STANDARDS-JAPANESE- RFA-Rankin_v7.5
- M-Appendix-M-0108-STANDARDS-KOREAN- RFA-Rankin_v7.5
- M-Appendix-M-0108-STANDARDS-LATVIAN- RFA-Rankin_v7.5
- M-Appendix-M-0108-STANDARDS-LITHUANIAN- RFA-Rankin_v7.5
- M-Appendix-M-0108-STANDARDS-MALAY- RFA-Rankin_v7.5
- M-Appendix-M-0108-STANDARDS-POLISH- RFA-Rankin_v7.5
- M-Appendix-M-0108-STANDARDS-PORTUGUESE- RFA-Rankin_v7.5
- M-Appendix-M-0108-STANDARDS-RUSSIAN- RFA-Rankin_v7.5
- M-Appendix-M-0108-STANDARDS-SERBIAN- RFA-Rankin_v7.5
- M-Appendix-M-0108-STANDARDS-SPANISH- RFA-Rankin_v7.5
- M-Appendix-M-0108-STANDARDS-SWEDISH- RFA-Rankin_v7.5
- M-Appendix-M-0108-STANDARDS-TAMIL- RFA-Rankin_v7.5
- M-Appendix-M-0108-STANDARDS-TURKISH- RFA-Rankin_v7.5
- M-Appendix-M-0108-STANDARDS-UKRAINIAN- RFA-Rankin_v7.5
- M-Appendix-M-0108-STANDARDS-VIETNAMESE- RFA-Rankin_v7.5
Action Research Arm Test (ARAT)
- M-Appendix-M-0108-STANDARDS-ENGLISH- ActionResearchArmTest-ARAT_v7.5
- M-Appendix-M-0108-STANDARDS-ARABIC- ActionResearchArmTest-ARAT_v7.5
- M-Appendix-M-0108-STANDARDS-BULGARIAN- ActionResearchArmTest-ARAT_v7.5
- M-Appendix-M-0108-STANDARDS-CHINESE TRADITIONAL- ActionResearchArmTest-ARAT_v7.5
- M-Appendix-M-0108-STANDARDS-CROATIAN- ActionResearchArmTest-ARAT_v7.5
- M-Appendix-M-0108-STANDARDS-CZECH- ActionResearchArmTest-ARAT_v7.5
- M-Appendix-M-0108-STANDARDS-DANISH- ActionResearchArmTest-ARAT_v7.5
- -Appendix-M-0108-STANDARDS-DUTCH- ActionResearchArmTest-ARAT_v7.5
- M-Appendix-M-0108-STANDARDS-FINNISH- ActionResearchArmTest-ARAT_v7.5
- M-Appendix-M-0108-STANDARDS-FRENCH- ActionResearchArmTest-ARAT_v7.5
- M-Appendix-M-0108-STANDARDS-GERMAN- ActionResearchArmTest-ARAT_v7.5
- M-Appendix-M-0108-STANDARDS-GREEK- ActionResearchArmTest-ARAT_v7.5
- M-Appendix-M-0108-STANDARDS-HEBREW- ActionResearchArmTest-ARAT_v7.5
- M-Appendix-M-0108-STANDARDS-HUNGARIAN- ActionResearchArmTest-ARAT_v7.5
- M-Appendix-M-0108-STANDARDS-ITALIAN- ActionResearchArmTest-ARAT_v7.5
- M-Appendix-M-0108-STANDARDS-JAPANESE- ActionResearchArmTest-ARAT_v7.5
- M-Appendix-M-0108-STANDARDS-KOREAN- ActionResearchArmTest-ARAT_v7.5
- M-Appendix-M-0108-STANDARDS-LATVIAN- ActionResearchArmTest-ARAT_v7.5
- M-Appendix-M-0108-STANDARDS-LITHUANIAN- ActionResearchArmTest-ARAT_v7.5
- M-Appendix-M-0108-STANDARDS-MALAY- ActionResearchArmTest-ARAT_v7.5
- M-Appendix-M-0108-STANDARDS-POLISH- ActionResearchArmTest-ARAT_v7.5
- M-Appendix-M-0108-STANDARDS-PORTUGUESE- ActionResearchArmTest-ARAT_v7.5
- M-Appendix-M-0108-STANDARDS-RUSSIAN- ActionResearchArmTest-ARAT_v7.5
- M-Appendix-M-0108-STANDARDS-SERBIAN- ActionResearchArmTest-ARAT_v7.5
- M-Appendix-M-0108-STANDARDS-SPANISH- ActionResearchArmTest-ARAT_v7.5
- M-Appendix-M-0108-STANDARDS-SWEDISH- ActionResearchArmTest-ARAT_v7.5
- M-Appendix-M-0108-STANDARDS-TAMIL- ActionResearchArmTest-ARAT_v7.5
- M-Appendix-M-0108-STANDARDS-TURKISH- ActionResearchArmTest-ARAT_v7.5
- M-Appendix-M-0108-STANDARDS-UKRAINIAN- ActionResearchArmTest-ARAT_v7.5
- M-Appendix-M-0108-STANDARDS-VIETNAMESE- ActionResearchArmTest-ARAT_v7.5
Gait Velocity (GV)
- M-Appendix-M-0108-STANDARDS-ENGLISH- GaitVelocity-GV_v7.5
- M-Appendix-M-0108-STANDARDS-ARABIC- GaitVelocity-GV_v7.5
- M-Appendix-M-0108-STANDARDS-BULGARIAN- GaitVelocity-GV_v7.5
- M-Appendix-M-0108-STANDARDS-CHINESE TRADITIONAL- GaitVelocity-GV_v7.5
- M-Appendix-M-0108-STANDARDS-CROATIAN- GaitVelocity-GV_v7.5
- M-Appendix-M-0108-STANDARDS-CZECH- GaitVelocity-GV_v7.5
- -Appendix-M-0108-STANDARDS-DANISH- GaitVelocity-GV_v7.5
- M-Appendix-M-0108-STANDARDS-DUTCH- GaitVelocity-GV_v7.5
- M-Appendix-M-0108-STANDARDS-FINNISH- GaitVelocity-GV_v7.5
- M-Appendix-M-0108-STANDARDS-FRENCH- GaitVelocity-GV_v7.5
- M-Appendix-M-0108-STANDARDS-GERMAN- GaitVelocity-GV_v7.5
- M-Appendix-M-0108-STANDARDS-GREEK- GaitVelocity-GV_v7.5
- M-Appendix-M-0108-STANDARDS-HEBREW- GaitVelocity-GV_v7.5
- M-Appendix-M-0108-STANDARDS-HUNGARIAN- GaitVelocity-GV_v7.5
- M-Appendix-M-0108-STANDARDS-ITALIAN- GaitVelocity-GV_v7.5
- M-Appendix-M-0108-STANDARDS-JAPANESE- GaitVelocity-GV_v7.5
- M-Appendix-M-0108-STANDARDS-KOREAN- GaitVelocity-GV_v7.5
- M-Appendix-M-0108-STANDARDS-LATVIAN- GaitVelocity-GV_v7.5
- M-Appendix-M-0108-STANDARDS-LITHUANIAN- GaitVelocity-GV_v7.5
- M-Appendix-M-0108-STANDARDS-MALAY- GaitVelocity-GV_v7.5
- M-Appendix-M-0108-STANDARDS-POLISH- GaitVelocity-GV_v7.5
- M-Appendix-M-0108-STANDARDS-PORTUGUESE- GaitVelocity-GV_v7.5
- M-Appendix-M-0108-STANDARDS-RUSSIAN- GaitVelocity-GV_v7.5
- M-Appendix-M-0108-STANDARDS-SERBIAN- GaitVelocity-GV_v7.5
- M-Appendix-M-0108-STANDARDS-SPANISH- GaitVelocity-GV_v7.5
- M-Appendix-M-0108-STANDARDS-SWEDISH- GaitVelocity-GV_v7.5
- M-Appendix-M-0108-STANDARDS-TAMIL- GaitVelocity-GV_v7.5
- M-Appendix-M-0108-STANDARDS-TURKISH- GaitVelocity-GV_v7.5
- M-Appendix-M-0108-STANDARDS-UKRAINIAN- GaitVelocity-GV_v7.5
- M-Appendix-M-0108-STANDARDS-VIETNAMESE- GaitVelocity-GV_v7.5
Motricity Index (MI)
- M-Appendix-M-0108-STANDARDS-ENGLISH- MotricityIndex-MI_v7.5
- M-Appendix-M-0108-STANDARDS-ARABIC- MotricityIndex-MI_v7.5
- M-Appendix-M-0108-STANDARDS-BULGARIAN- MotricityIndex-MI_v7.5
- M-Appendix-M-0108-STANDARDS-CHINESE TRADITIONAL- MotricityIndex-MI_v7.5
- -Appendix-M-0108-STANDARDS-CROATIAN- MotricityIndex-MI_v7.5
- M-Appendix-M-0108-STANDARDS-CZECH- MotricityIndex-MI_v7.5
- M-Appendix-M-0108-STANDARDS-DANISH- MotricityIndex-MI_v7.5
- M-Appendix-M-0108-STANDARDS-DUTCH- MotricityIndex-MI_v7.5
- M-Appendix-M-0108-STANDARDS-FINNISH- MotricityIndex-MI_v7.5
- M-Appendix-M-0108-STANDARDS-FRENCH- MotricityIndex-MI_v7.5
- M-Appendix-M-0108-STANDARDS-GERMAN- MotricityIndex-MI_v7.5
- M-Appendix-M-0108-STANDARDS-GREEK- MotricityIndex-MI_v7.5
- M-Appendix-M-0108-STANDARDS-HEBREW- MotricityIndex-MI_v7.5
- M-Appendix-M-0108-STANDARDS-HUNGARIAN- MotricityIndex-MI_v7.5
- M-Appendix-M-0108-STANDARDS-ITALIAN- MotricityIndex-MI_v7.5
- M-Appendix-M-0108-STANDARDS-JAPANESE- MotricityIndex-MI_v7.5
- M-Appendix-M-0108-STANDARDS-KOREAN- MotricityIndex-MI_v7.5
- M-Appendix-M-0108-STANDARDS-LATVIAN- MotricityIndex-MI_v7.5
- M-Appendix-M-0108-STANDARDS-LITHUANIAN- MotricityIndex-MI_v7.5
- M-Appendix-M-0108-STANDARDS-MALAY- MotricityIndex-MI_v7.5
- M-Appendix-M-0108-STANDARDS-POLISH- MotricityIndex-MI_v7.5
- M-Appendix-M-0108-STANDARDS-PORTUGUESE- MotricityIndex-MI_v7.5
- M-Appendix-M-0108-STANDARDS-RUSSIAN- MotricityIndex-MI_v7.5
- M-Appendix-M-0108-STANDARDS-SERBIAN- MotricityIndex-MI_v7.5
- M-Appendix-M-0108-STANDARDS-SPANISH- MotricityIndex-MI_v7.5
- M-Appendix-M-0108-STANDARDS-SWEDISH- MotricityIndex-MI_v7.5
- M-Appendix-M-0108-STANDARDS-TAMIL- MotricityIndex-MI_v7.5
- M-Appendix-M-0108-STANDARDS-TURKISH- MotricityIndex-MI_v7.5
- M-Appendix-M-0108-STANDARDS-UKRAINIAN- MotricityIndex-MI_v7.5
- M-Appendix-M-0108-STANDARDS-VIETNAMESE- MotricityIndex-MI_v7.5
Enhancing Participation in Clinical Trials — Eligibility Criteria, Enrollment Practices, and Trial Designs (FDA Guidance)
Additional Industry Guidances
- FDA 2025 Investigations Operations Manual (IOM)
- FDA Remote Regulatory Assessments (RRAs)
- WHO Guidance for Best Practices for Clinical Trials
- Global Visionaries, Compliance, Privacy and Scientific Key Opinion Leaders (KOLs) Video Interviews
- European Medicines Agency (EMA) – Clinical Trial Information System (CTIS) User Guidance on the Sponsor’s Workspace